Synthesis of β-Amino Alcohol Derivatives through a Photo-Induced Reaction in DMSO
Synthetic routes to β-amino alcohols and their derivatives are extremely important as these compounds are widespread in natural products, pharmaceuticals, and agrochemicals. In 2018, Yu’s group published an interesting photoredox-induced radical relay method in DMSO to synthesize functionalized β-amino alcohol derivatives.1 Unlike other established methods to simultaneously construct a C-N and a C-O bond across the carbon-carbon double bond of an alkene, this new method is highly regioselective. The benefits of this new method also include the fact that it is light-induced (white LED lights) and therefore is run under mild conditions with 2 mol % of an iridium catalyst. Again, DMSO is shown to be the preferred solvent for a light-induced reaction. (For other recent examples, see previous Synthesis Corner posts from August 2, 2018, and July 9, 2018.)
Starting with an O-acyl hydroxylamine 1 and a vinyl ether 2, this synthetic strategy was found to be effective in producing 1,2-amino alcohol derivatives from either an arylacetylene 3 or a styrene 4, resulting in 3-alkenyl-1-amino-2-ols 5 and 4-oxo-1-amino-2-ol derivatives 6, respectively (Figure 1).
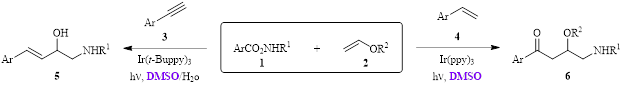
Figure 1. Light-induced reactions to produce functionalized β-Amino Alcohol Derivatives
After optimization of both reactions, the substrate scope was investigated. In the reaction with phenylacetylenes 3 (Figure 2), the reaction was tested with a variety of substituents appended to the phenyl ring at various positions. All of these provided moderate to good isolated yields, ranging from 46–91% (15 examples). The reaction was also tolerant of larger aromatic systems and heteroaromatic rings, giving yields between 49–72% (4 examples).
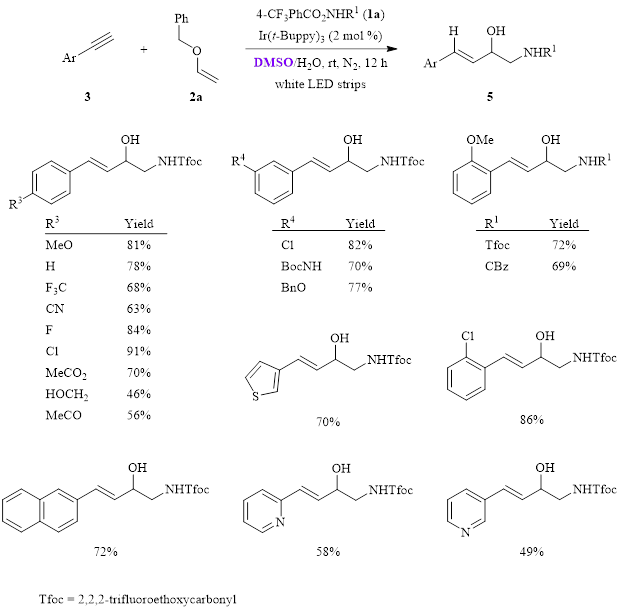
Figure 2. Substrate scope study for the reaction using phenylacetylenes 3
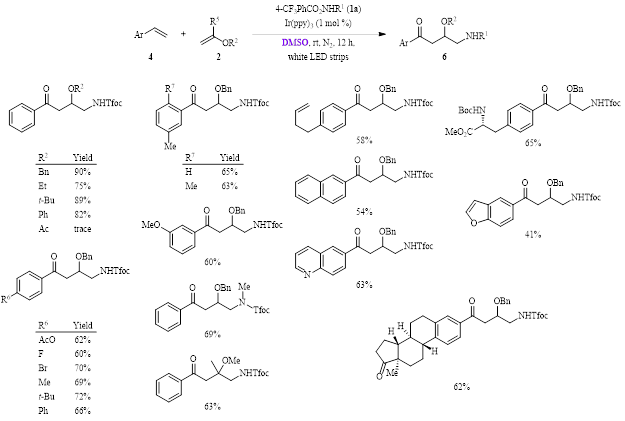
When using different styrenes 4 (Figure 3), it can be seen that the reaction again tolerates a variety of substituents on both the styrene and the vinyl ether 2. These yields range from 41–90% (21 examples), with only vinyl acetate giving a low yield (trace amount).
Both reactions in Figure 2 and 3 are thought to proceed through a radical pathway whereby the O-acyl hydroxylamine produces a nitrogen radical that reacts with the vinyl ether. This produces a carbon radical that is adjacent to the oxygen atom. This step provides the 1,2-amino alcohol pattern. This carbon radical then adds to the unsaturated aryl compounds (3 or 4) to provide the carbon skeleton of the product. It is thought that the 4-oxo product 6 ultimately results from a Kornblum oxidation. Therefore, DMSO also acts as a reactant in the reaction pathway involving the various styrenes.
In summary, this group has developed a novel one pot radical reaction strategy targeting 1,2-amino alcohols from various styrenes and arylacetylenes. Because this is a photo-induced reaction, the conditions are mild and amenable to a wide variety of substrates. Again, DMSO is found to be the preferred solvent for this light promoted reaction.
Debra D. Dolliver, Ph.D.
1 An, W.-D.; Jiao, Y.-Y.; Zhang, H.; Gao, Y.; Yu, S. Org. Lett. 2018, 20, 401.

Leave a Reply
You must be logged in to post a comment.