DMSO as the Solvent for Cu(I)-Catalyzed Reactions: The Synthesis of Pyrrole Derivatives
Recently, Zhao’s group reported on the oxidative cyclization of maleimides with amines and alkyne esters to form pyrrole and dihydropyrrole derivatives (Figure 1).1 The importance of these five-membered nitrogen-containing heterocyclic compounds cannot be overstated, as they are common in biologically- and pharmaceutically-active compounds. Therefore, the chemical literature is filled with examples of routes to form these nitrogen heterocycles and to functionalize them once formed. The work by Zhao’s group is innovative in that it efficiently builds these highly functionalized nitrogen heterocycles via a route using simple building blocks (1° amines, ester acetylenes, and maleimides) and a cheap, readily-available, and relatively green catalyst (CuCl) in DMSO.
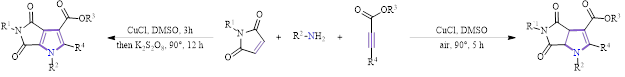
Figure 1: General scheme for the Cu(I)-catalyzed synthesis of functionalized pyrroles and dihydropyrroles
During the optimization of the Cu(I)-catalyzed reaction for the formation of the functionalized dihydropyrroles, DMSO was found to be superior to all other solvents tested (DMF, DCM, ACN, DMA, dioxane, HOAc, toluene, DCE, and PhCl). In other words, use of DMSO as the solvent led to significantly higher yields. This adds to the growing number of Cu(I) catalyzed reactions where DMSO either plays a role in the reaction (see Jan 18, 2018 Synthesis Corner post) or acts as the best solvent for the reaction (see May 30, 2018 Synthesis Corner post).
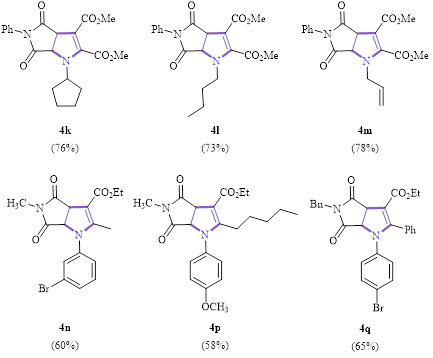
A substrate study for the synthesis of the dihydropyrroles using the optimized conditions is shown in Scheme 1. As can be seen, all reactions had >50% yield (except in the case of the unsubstituted maleimide, 4e, R4 = H, 40% yield). The reaction tolerated a variety of groups at various positions: electron-donating, electron-withdrawing, aryl, alkyl, and alkenyl.
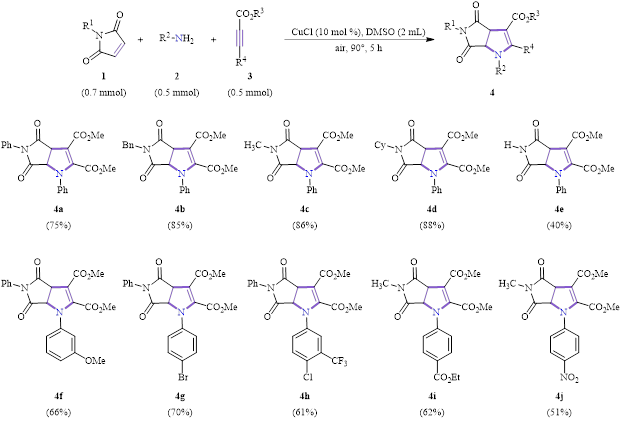
Scheme 1: Substrate scope for the synthesis of dihydropyrans
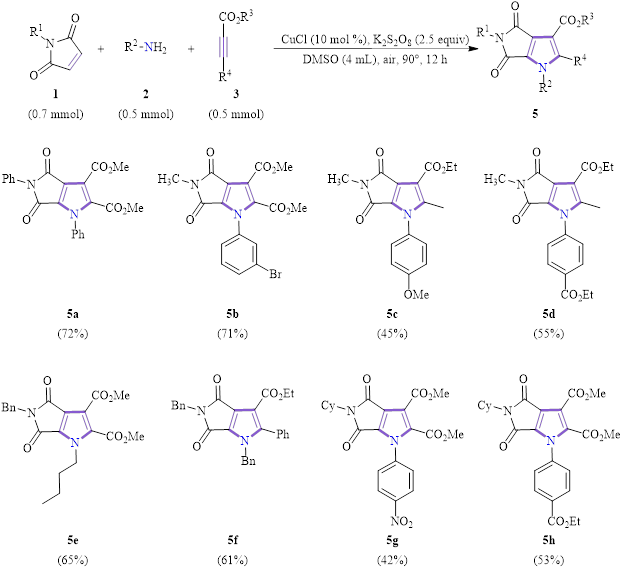
These researchers found that the inclusion of an oxidant in the above reaction conditions led to a dehydrogenation aromatization reaction to yield valuable fully-substituted pyrroles. This reaction was also tested for substrate scope, and the optimized conditions and results with a variety of substituents at various locations are shown in Scheme 2.
Scheme 2: Substrate scope for the synthesis of functionalized pyrroles.
This reaction also showed good tolerance for electron-withdrawing, electron-donating, aryl and alkyl substitutents, giving moderate to good yields in all examples.
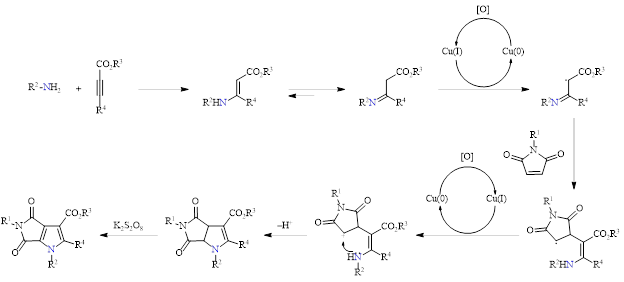
A series of experiments including TEMPO in the reaction were performed to probe the mechanism, and this indicated that the reaction proceeds through a radical process. Based on this and other reports in the literature, these investigators proposed the following mechanism (Scheme 3).
Scheme 3: Proposed mechanism.
In conclusion, the Zhao group has developed a Cu(I)-catalyzed route to highly-functionalized dihydropyrroles and pyrroles using DMSO as the solvent. This reaction occurs under mild conditions and with readily available starting materials and catalyst.
Debra D. Dolliver, Ph.D.
References:
- Zhu, J.-N.; Chen, L.-L.; Zhou, R.-X.; Li, B.; Shao, Z.-Y.; Zhao, S.-Y. Lett. 2017, 19, 6044-6047.







Leave a Reply
You must be logged in to post a comment.