Gaylord Chemical specializes in the production of DMSO and DMS, which can be used in a variety of markets. Discover more about the meaning of DMSO and DMS below.
DMSO
Dimethyl Sulfoxide (DMSO) is a highly polar and water miscible organic liquid. It is essentially odorless and has a low level of toxicity. As do all dipolar aprotic solvents, DMSO has a relatively high boiling point. View more details on the chemical structure of DMSO and contact us for more information.
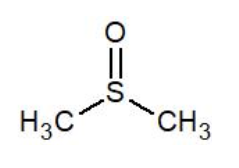
| DMSO Properties | |
| DMSO Chemical Formula | C₂H₆OS |
| Molecular weight | 78.13 g/mol |
| CAS No. | [67-68-5] |
| Appearance | Colorless liquid |
| Specific Gravity | 1.1 (20°C) |
| Melting point | 18°C (66°F) |
| Boiling point | 189°C (372°F) |
| Solubility in water | Miscible |
| Acidity (pKa) | 35 |
| Flash Point | 95°C (open cup) |
| Viscosity | 2.0 cP (25°C) |
Applications for Dimethyl Sulfoxide
A superior reaction solvent for the production of Active Pharmaceutical Ingredients and intermediates. Many specific reactions run best in DMSO, producing higher product yields. The high polarity of DMSO allows it to dissolve many compounds that other solvents cannot. DMSO is itself a reactant in a class of environmentally-friendly oxidation reactions.
A component of microelectronic cleaning formulations. A DMSO product grade containing trace metals levels is important in surface cleaning operations involved with microelectronic device production. DMSO is used in lithographic cleaning steps as a photoresist stripper and in Cu post-etch residue removal (PERR) products.
As a cryopreservation agent. DMSO has the unusual ability to depress the freezing point of water dramatically, while maintaining viability with many cell and tissue types. This has led to its use in cryopreservation media for stem cells derived from human umbilical cord blood and reproductive tissue.
As a production and processing solvent for polymers. DMSO is used to produce carbon fiber and certain high-performance polymers. As it can dissolve many polymers DMSO can be used to cast polymeric films and fibers useful in the manufacture of purification membranes. DMSO can be used to clean polymeric residue(polyurethane/polyester) from parts manufacturing.
DMS
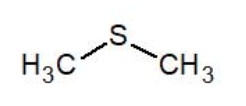
Dimethyl Sulfide (DMS) is a low molecular weight sulfur compound with a distinctive garlic-like odor. It possesses a low level of toxicity and is used as a flavor component in the US and other parts of the world. View more details on DMS structure below and contact us for more information.
Safety Data Sheets
| DMS Properties | |
| Molecular formula | C₂H₆S |
| Molecular weight | 78.13 g/mol |
| CAS No. | [78-18-3] |
| Melting point | -98.3°C (-145°F) |
| Boiling point | 37°C (98.6°F) |
| Solubility in water | Max 2% w/v (25°C) |
| Flash Point | -48°C (-54°F) |
Key Applications for Dimethyl Sulfide
As an effective presulfiding agent. DMS is used as a sulfur source for industrial catalysts used in hydrodesulfurization, hydrodenitrification, and catalytic cracking. These catalysts are supplied in their oxide form, and must be modified with sulfur prior to use
As a furnace passivation agent. The service lifetime of pyrolysis furnace tubes used to manufacture ethylene is extended when treated with low levels of DMS. Furnace tubes used to reduce iron ore are protected in a similar way.
As a “back-up” odorant in gas products. DMS is blended with common gas odorants as a protective measure. When odorants such as methyl mercaptan oxidize, their odor diminishes. DMS is not as susceptible to this problem.
In chemical synthesis, DMS is alkylated to produce trimethylsulfonium halides, which are useful epoxidation reagents. DMS-BH3 is used in hydroboration-oxidation and related reactions.







