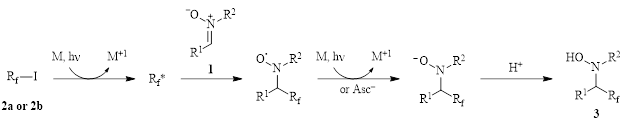
Figure 1: Photocatalyzed addition of fluoroalkyl groups to nitrones in DMSO.
Photocatalysis in DMSO: Reductive Fluoroalkylation of Nitrones

The yields for this reaction are generally good to excellent with the only yield below 50% occurring when R2 = t-butyl group.
A major development with this work is the ability to add partially fluorinated building blocks (2b) to the C=N double bond. This type of addition is unavailable through carbanionic fluoroalkylation methods due to facile b-elimination in these species under these conditions.
This method is thought to proceed through the addition of fluorinated radicals to the C=N double bond, although reactions performed in the presence of TEMPO were inconclusive as ascorbic acid is able to reduce TEMPO to TEMPO-H. Therefore, the authors examined the reduction potentials of the catalyst and the reagents and concluded that a pathway involving the reductive quenching of the photocatalyst by ascorbic acid seemed most likely. This led them to propose the mechanism shown in Figure 2. Here, the fluoroalkyl iodide is reduced by the photocatalyst to generate the fluoroalkyl radical which adds to the nitrone 1. Another single electron transfer and subsequent protonation produces the hydroxylamine product 3.
Figure 2: Proposed mechanism
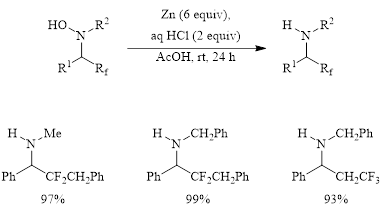
Dilman et al. also investigated the reduction of the fluorinated hydroxylamines to amines. They found that these fluorinated hydroxylamines were easily reduced to the corresponding amine with excellent yields (Figure 3).
Figure 3: Conversion of fluorinated hydroxylamines to amines.
In general, this is a gentle method to perform radical fluoroalkylations at the C=N double bond through a photocatalytic route, leading to valuable fluorinated hydroxylamines and amines. Using DMSO as the solvent leads to the best yields. This work represents a way to introduce not only perfluorinated species but also partially fluorinated species. This is a substantial advance as the addition of these partially fluorinated groups is not available through the traditional carbanionic fluoroalkylation route.
Debra D. Dolliver, Ph. D.
1Supranovich, V. I.; Levin, V. V.; Struchkova, M. I.; Dilman, A. D. Org. Lett. 2018, 20, 840–843