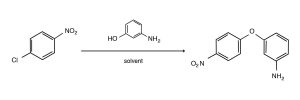
SNAr Comparative Reaction
Uses:
Nucleophilic Aromatic Substitution (SNAr) reactions are essential tools for the synthesis of many pharmaceutical targets (e.g. quinolone antibiotics) and agricultural products such as the dinitrophenyl ether herbicides.
Advantages:
The essential advantage of DMSO in these reactions is that due to the lack of solvation of anions in this solvent. These naked species are extremely reactive in DMSO. Thus, rate enhancements of SNAr reactions in DMSO of the order of 106 x have been reported. Consequently, SNAr reactions in DMSO can be performed at lower temperatures than in conventional solvents, including other dipolar aprotic solvents. Inasmuch as side-reactions in DMSO are minimized, this translates to selectivity advantages in DMSO relative to conventional solvents.
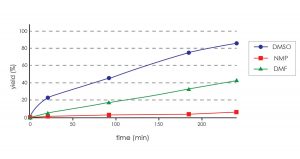
It is clear from the graph below that reactions in DMSO are faster than the comparable reactions run with other solvents. Increasing the temperature to accelerate the rate of product formation in other solvents often results in the formation of unwanted side reactions.
Contact Gaylord Chemical!
Gaylord Chemical can provide product samples to qualified users of chemical products. Request a Sample Today!