Condensation reactions may be intermolecular or intramolecular and typically involve the elimination of water or some other simple molecule. Many of the ordinary reactions of carbonyl compounds can be accomplished in DMSO. Good results can be obtained due to the greater solubility of reactants or because of the enhanced reactivity of nucleophiles.
The esters of carboxylic acids react with the dimsyl ion in DMSO to yield ß-keto sulfoxides [Martin, D.; Niclas, H-J., Ber. 102, 31-37 (1969); Shilling, W. L., Crown Zellerbach, results unpublished].
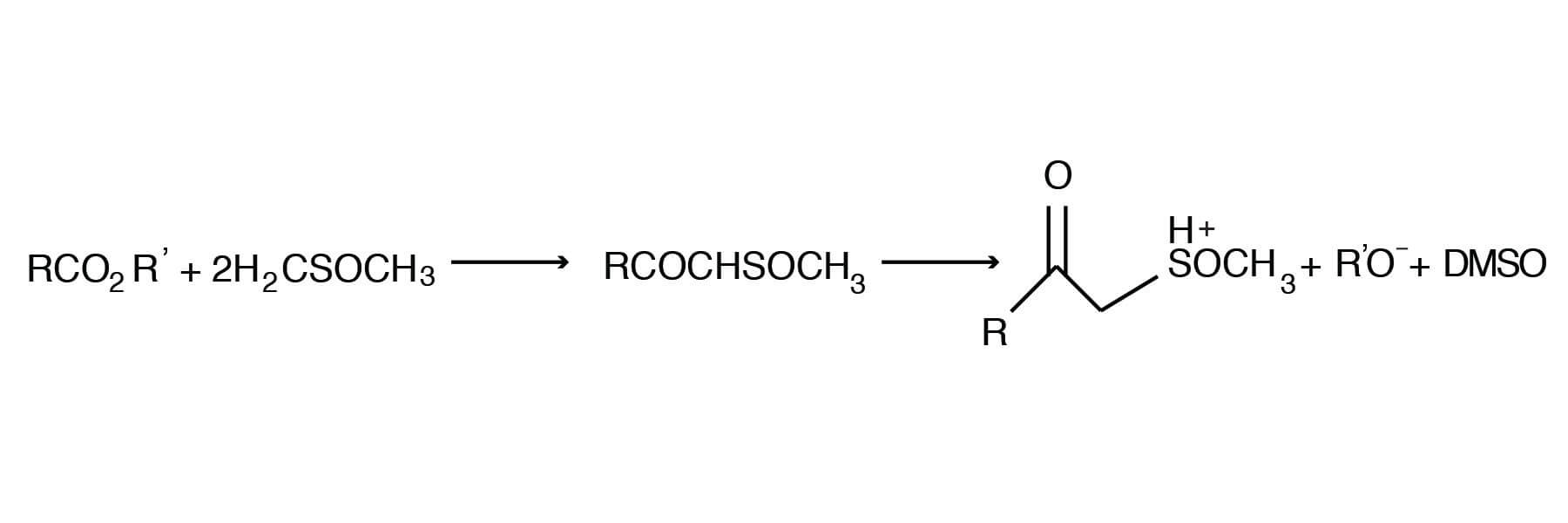
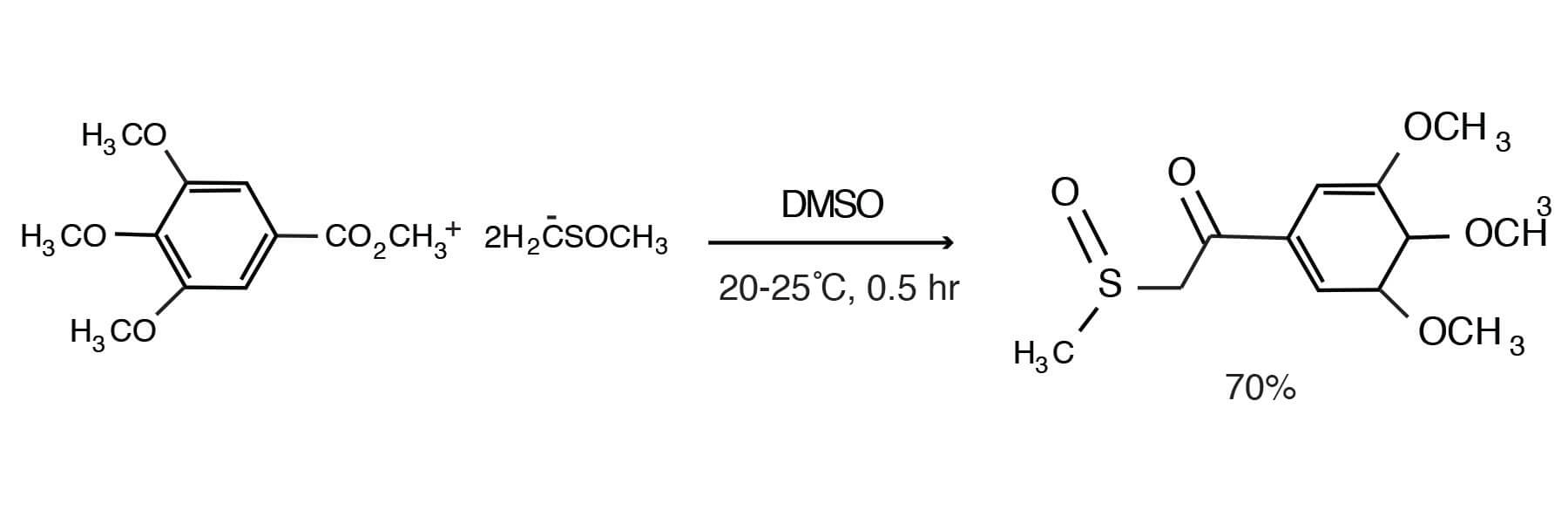
This condensation reaction has found application in the synthesis of useful intermediates. Thus, a number of benzoic acid esters can be reacted with the dimsyl ion in DMSO to give the corresponding ß-keto sulfoxides [Cresswell, R. M.; Mentha, J. W., U.S. 3,878,252 (April 15, 1975); CA 83, 78855R].