Introduction
Dimethyl sulfide (DMS) is a low molecular weight, organosulfur compound that is produced industrially, while also being commonly found in nature. In fact, oceanic dimethyl sulfide is the dominant natural source for sulfur in the atmosphere (Bates et al., 1992; Gondwe et al., 2003). The low toxicity level in this compound is further exemplified by its use as a synthetic flavoring substance and adjuvant (CFR 21§172.515) in the United States.
Contents
- Physical & Chemical Properties
- Vapor Pressure Temperature
- Petrochemical Applications
- Fine Chemical Synthesis
- Polymer Applications
- Life Science Applications
- Storage and Handling
DMS synonyms: Methane, thiobis-; Methyl sulfide; Dimethyl monosulfide; Dimethyl thioether; DMS; Methyl Monosulfide; 2-Thiapropane; Dimethyl sulphide; Thiobismethane; (CH3)2S; Dimethylsulfide; Exact-S; Methyl sulphide; Methylthiomethane; Sulfure de methyle; 2-Thiopropane; UN 1164; Methylthioether; Thiopropane; Sulfide, methyl-; (Methylsulfanyl)methane
Autoignition Temperature (ASTM E 659): 206°C (403°F)
Boiling Point: 38°C (99°F)
Boiling Range (Anhydrous): 90% @37.1 +0.2° C
CAS Registry Number: 75-18-3
Coefficient of Expansion: 0. 0013 ml/ml/°C.
Critical Pressure: 826 psia= 56.1 atm= 5.69 MN/sq m
[U.S. Coast Guard, Department of Transportation. CHRIS – Hazardous Chemical Data. Manual Two. Washington, DC: U.S. Government Printing Office, Oct., 1978.]
Critical Temperature: 444 deg F= 229 deg C= 502 deg K Density/Specific Gravity: 0.847 (20°C) (water = 1) Dielectric Constant: 12
Evaporation rate (n-butyl acetate = 1): »1.0
Flash Point (ASTM D-93, closed cup): -48° C (-54° F)
Flash Point (calculated): -34° C
Formula: C2H6S
Freezing Point: -98.3°C (-145°F)
Hansen Solubility Data:
Dispersion 16.1
Polar 6.4
Hydrogen Bonding 7.4
Heat of Combustion: 13,200 Btu/lb= 7340 cal/g= 457.3 K cal/mol= 307X10+5 J/kg
[U.S. Coast Guard, Department of Transportation. CHRIS – Hazardous Chemical Data. Manual Two. Washington, DC: U.S. Government Printing Office, Oct., 1978.]
Heat of Fusion: 30.73 cal/g
Heat of Vaporization: 194 Btu/lb= 108 cal/g= 4.52X10+5 J/kg
[U.S. Coast Guard, Department of Transportation. CHRIS – Hazardous Chemical Data. Manual Two. Washington, DC: U.S. Government Printing Office, Oct., 1978.]
Henry’ Law Constant: 1.61×10-3 atm cu-m/mol-3 @ 25° C
[Gaffney JS et al; Environ Sci Technol 21: 519-23 (1987)]
Hydroxyl radical rate constant: 4.56×10-12 cu-cm/molc sec @ 25° C
[Atkinson R; Journal ofPhysical And Chemical Reference Data. Monograph No 1 (1989)]
Interfacial Tension, Liquid-Water: (est) 30 dynes/cm= 0.030 N/m @ 20° C; Ratio of Specific Heats of Vapor (Gas): 1.1277 @ 16°C
[U.S. Coast Guard, Department of Transportation. CHRIS – Hazardous Chemical Data. Manual Two. Washington, DC: U.S. Government Printing Office, Oct., 1978]
IR: 6300 (Sadtler Research Laboratories Prism Collection)
[Weast, R.C. and M.J. Astle. CRC Handbook of Data on Organic Compounds. Volumes I and II. Boca Raton, FL: CRC Press Inc. 1985., p. V1 853]
Lower Explosive Limit: 2.2%
MASS: 10 (National Bureau of Standards EPA-NIH Mass Spectra Data Base, NSRDS-NBS-63)
[Weast, R.C. and M.J. Astle. CRC Handbook of Data on Organic Compounds. Volumes I and II. Boca Raton, FL: CRC Press Inc. 1985., p. V1 853]
Molar Volume: 73.2
Molecular Weight: 62.14
Octanol/Water Partition Coefficient: 0.92
Odor Threshold: less than 1 ppm
Odor: Unpleasant odor of wild radish or cabbage
[Fenaroli’s Handbook of Flavor Ingredients. Volume 2. Edited, translated, and revised by T.E. Furia and N. Bellanca. 2nd ed. Cleveland: The Chemical Rubber Co., 1975., p. 406]
pH: not available
Physical form: Colorless Liquid
Refractive Index: 1.4355 @ 20° C/D; MAX ABSORPTION (ALCOHOL): 215 NM (LOG E= 3.0)
[Weast, R.C. (ed.). Handbook of Chemistry and Physics. 60th ed. Boca Raton, Florida: CRC Press Inc., 1979, p. C-505]
Solubility in Water: Max. 2% (w/v) @ 25° C
Specific Heat: @ 20° C Cp (Cal/gm/°C)=0.45
Sulfur Content: 51.5%
Surface Tension: 26.5 dynes/cm= 0.0265 N/m @ 11 deg C 23.33 dynes/cm @ 32. 9° C 19. 87 dynes/cm@ 57. 7° C
[U.S. Coast Guard, Department of Transportation. CHRIS – Hazardous Chemical Data. Manual Two. Washington, DC: U.S. Government Printing Office, Oct., 1978.]
Upper Explosive Limit: 19.7%
Vapor Density: 2.14 (Air= 1)
[Sax, N.I. Dangerous Properties of Industrial Materials. 5th ed. New York: Van Nostrand Rheinhold, 1979., p. 832]
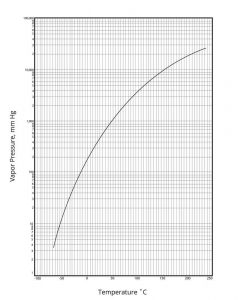
Vapor Pressure @ 20°C (68°F): 530 mbar (398 mm Hg)
Vapor Pressure: 502 mm Hg @ 25° C , from experimentally-derived coefficients.
[Daubert, T.E., R.P. Danner. Physical and Thermodynamic Properties of Pure Chemicals Data Compilation. Washington, D.C.: Taylor and Francis, 1989.]
Viscosity at 25°C (68°F): 0.29 cps
Viscosity: Less than 32.6 Saybolt unit sec
[Clayton, G. D. and F. E. Clayton (eds.). Patty’s Industrial Hygiene and Toxicology: Volume 2A, 2B, 2C: Toxicology. 3rd ed. New York: John Wiley Sons, 1981-1982., p. 2065]
Water Soluble in DMS: Max. 0.4% (w/v) @ 25° C
Wt/vol conversion: 2.54 mg/cu m = 1 ppm
[Clayton, G. D. and F. E. Clayton (eds.). Patty’s Industrial Hygiene and Toxicology: Volume 2A, 2B, 2C: Toxicology. 3rd ed. New York: John Wiley Sons, 1981-1982., p. 2065]
DMS synonyms: Methane, thiobis-; Methyl sulfide; Dimethyl monosulfide; Dimethyl thioether; DMS; Methyl Monosulfide; 2-Thiapropane; Dimethyl sulphide; Thiobismethane; (CH3)2S; Dimethylsulfide; Exact-S; Methyl sulphide; Methylthiomethane; Sulfure de methyle; 2-Thiopropane; UN 1164; Methylthioether; Thiopropane; Sulfide, methyl-; (Methylsulfanyl)methane
Autoignition Temperature (ASTM E 659): 206°C (403°F)
Boiling Point: 38°C (99°F)
Boiling Range (Anhydrous): 90% @37.1 +0.2° C
CAS Registry Number: 75-18-3
Coefficient of Expansion: 0. 0013 ml/ml/°C.
Critical Pressure: 826 psia= 56.1 atm= 5.69 MN/sq m
[U.S. Coast Guard, Department of Transportation. CHRIS – Hazardous Chemical Data. Manual Two. Washington, DC: U.S. Government Printing Office, Oct., 1978.]
Critical Temperature: 444 deg F= 229 deg C= 502 deg K Density/Specific Gravity: 0.847 (20°C) (water = 1) Dielectric Constant: 12
Evaporation rate (n-butyl acetate = 1): »1.0
Flash Point (ASTM D-93, closed cup): -48° C (-54° F)
Flash Point (calculated): -34° C
Formula: C2H6S
Freezing Point: -98.3°C (-145°F)
Hansen Solubility Data:
Dispersion 16.1
Polar 6.4
Hydrogen Bonding 7.4
Heat of Combustion: 13,200 Btu/lb= 7340 cal/g= 457.3 K cal/mol= 307X10+5 J/kg
[U.S. Coast Guard, Department of Transportation. CHRIS – Hazardous Chemical Data. Manual Two. Washington, DC: U.S. Government Printing Office, Oct., 1978.]
Heat of Fusion: 30.73 cal/g
Heat of Vaporization: 194 Btu/lb= 108 cal/g= 4.52X10+5 J/kg
[U.S. Coast Guard, Department of Transportation. CHRIS – Hazardous Chemical Data. Manual Two. Washington, DC: U.S. Government Printing Office, Oct., 1978.]
Henry’ Law Constant: 1.61×10-3 atm cu-m/mol-3 @ 25° C
[Gaffney JS et al; Environ Sci Technol 21: 519-23 (1987)]
Hydroxyl radical rate constant: 4.56×10-12 cu-cm/molc sec @ 25° C
[Atkinson R; Journal ofPhysical And Chemical Reference Data. Monograph No 1 (1989)]
Interfacial Tension, Liquid-Water: (est) 30 dynes/cm= 0.030 N/m @ 20° C; Ratio of Specific Heats of Vapor (Gas): 1.1277 @ 16°C
[U.S. Coast Guard, Department of Transportation. CHRIS – Hazardous Chemical Data. Manual Two. Washington, DC: U.S. Government Printing Office, Oct., 1978]
IR: 6300 (Sadtler Research Laboratories Prism Collection)
[Weast, R.C. and M.J. Astle. CRC Handbook of Data on Organic Compounds. Volumes I and II. Boca Raton, FL: CRC Press Inc. 1985., p. V1 853]
Lower Explosive Limit: 2.2%
MASS: 10 (National Bureau of Standards EPA-NIH Mass Spectra Data Base, NSRDS-NBS-63)
[Weast, R.C. and M.J. Astle. CRC Handbook of Data on Organic Compounds. Volumes I and II. Boca Raton, FL: CRC Press Inc. 1985., p. V1 853]
Molar Volume: 73.2
Molecular Weight: 62.14
Octanol/Water Partition Coefficient: 0.92
Odor Threshold: less than 1 ppm
Odor: Unpleasant odor of wild radish or cabbage
[Fenaroli’s Handbook of Flavor Ingredients. Volume 2. Edited, translated, and revised by T.E. Furia and N. Bellanca. 2nd ed. Cleveland: The Chemical Rubber Co., 1975., p. 406]
pH: not available
Physical form: Colorless Liquid
Refractive Index: 1.4355 @ 20° C/D; MAX ABSORPTION (ALCOHOL): 215 NM (LOG E= 3.0)
[Weast, R.C. (ed.). Handbook of Chemistry and Physics. 60th ed. Boca Raton, Florida: CRC Press Inc., 1979, p. C-505]
Solubility in Water: Max. 2% (w/v) @ 25° C
Specific Heat: @ 20° C Cp (Cal/gm/°C)=0.45
Sulfur Content: 51.5%
Surface Tension: 26.5 dynes/cm= 0.0265 N/m @ 11 deg C 23.33 dynes/cm @ 32. 9° C 19. 87 dynes/cm@ 57. 7° C
[U.S. Coast Guard, Department of Transportation. CHRIS – Hazardous Chemical Data. Manual Two. Washington, DC: U.S. Government Printing Office, Oct., 1978.]
Upper Explosive Limit: 19.7%
Vapor Density: 2.14 (Air= 1)
[Sax, N.I. Dangerous Properties of Industrial Materials. 5th ed. New York: Van Nostrand Rheinhold, 1979., p. 832]
Vapor Pressure @ 20°C (68°F): 530 mbar (398 mm Hg)
Vapor Pressure: 502 mm Hg @ 25° C , from experimentally-derived coefficients.
[Daubert, T.E., R.P. Danner. Physical and Thermodynamic Properties of Pure Chemicals Data Compilation. Washington, D.C.: Taylor and Francis, 1989.]
Viscosity at 25°C (68°F): 0.29 cps
Viscosity: Less than 32.6 Saybolt unit sec
[Clayton, G. D. and F. E. Clayton (eds.). Patty’s Industrial Hygiene and Toxicology: Volume 2A, 2B, 2C: Toxicology. 3rd ed. New York: John Wiley Sons, 1981-1982., p. 2065]
Water Soluble in DMS: Max. 0.4% (w/v) @ 25° C
Wt/vol conversion: 2.54 mg/cu m = 1 ppm
[Clayton, G. D. and F. E. Clayton (eds.). Patty’s Industrial Hygiene and Toxicology: Volume 2A, 2B, 2C: Toxicology. 3rd ed. New York: John Wiley Sons, 1981-1982., p. 2065]
DMS synonyms: Methane, thiobis-; Methyl sulfide; Dimethyl monosulfide; Dimethyl thioether; DMS; Methyl Monosulfide; 2-Thiapropane; Dimethyl sulphide; Thiobismethane; (CH3)2S; Dimethylsulfide; Exact-S; Methyl sulphide; Methylthiomethane; Sulfure de methyle; 2-Thiopropane; UN 1164; Methylthioether; Thiopropane; Sulfide, methyl-; (Methylsulfanyl)methane
Autoignition Temperature (ASTM E 659): 206°C (403°F)
Boiling Point: 38°C (99°F)
Boiling Range (Anhydrous): 90% @37.1 +0.2° C
CAS Registry Number: 75-18-3
Coefficient of Expansion: 0. 0013 ml/ml/°C.
Critical Pressure: 826 psia= 56.1 atm= 5.69 MN/sq m
[U.S. Coast Guard, Department of Transportation. CHRIS – Hazardous Chemical Data. Manual Two. Washington, DC: U.S. Government Printing Office, Oct., 1978.]
Critical Temperature: 444 deg F= 229 deg C= 502 deg K Density/Specific Gravity: 0.847 (20°C) (water = 1) Dielectric Constant: 12
Evaporation rate (n-butyl acetate = 1): »1.0
Flash Point (ASTM D-93, closed cup): -48° C (-54° F)
Flash Point (calculated): -34° C
Formula: C2H6S
Freezing Point: -98.3°C (-145°F)
Hansen Solubility Data:
Dispersion 16.1
Polar 6.4
Hydrogen Bonding 7.4
Heat of Combustion: 13,200 Btu/lb= 7340 cal/g= 457.3 K cal/mol= 307X10+5 J/kg
[U.S. Coast Guard, Department of Transportation. CHRIS – Hazardous Chemical Data. Manual Two. Washington, DC: U.S. Government Printing Office, Oct., 1978.]
Heat of Fusion: 30.73 cal/g
Heat of Vaporization: 194 Btu/lb= 108 cal/g= 4.52X10+5 J/kg
[U.S. Coast Guard, Department of Transportation. CHRIS – Hazardous Chemical Data. Manual Two. Washington, DC: U.S. Government Printing Office, Oct., 1978.]
Henry’ Law Constant: 1.61×10-3 atm cu-m/mol-3 @ 25° C
[Gaffney JS et al; Environ Sci Technol 21: 519-23 (1987)]
Hydroxyl radical rate constant: 4.56×10-12 cu-cm/molc sec @ 25° C
[Atkinson R; Journal ofPhysical And Chemical Reference Data. Monograph No 1 (1989)]
Interfacial Tension, Liquid-Water: (est) 30 dynes/cm= 0.030 N/m @ 20° C; Ratio of Specific Heats of Vapor (Gas): 1.1277 @ 16°C
[U.S. Coast Guard, Department of Transportation. CHRIS – Hazardous Chemical Data. Manual Two. Washington, DC: U.S. Government Printing Office, Oct., 1978]
IR: 6300 (Sadtler Research Laboratories Prism Collection)
[Weast, R.C. and M.J. Astle. CRC Handbook of Data on Organic Compounds. Volumes I and II. Boca Raton, FL: CRC Press Inc. 1985., p. V1 853]
Lower Explosive Limit: 2.2%
MASS: 10 (National Bureau of Standards EPA-NIH Mass Spectra Data Base, NSRDS-NBS-63)
[Weast, R.C. and M.J. Astle. CRC Handbook of Data on Organic Compounds. Volumes I and II. Boca Raton, FL: CRC Press Inc. 1985., p. V1 853]
Molar Volume: 73.2
Molecular Weight: 62.14
Octanol/Water Partition Coefficient: 0.92
Odor Threshold: less than 1 ppm
Odor: Unpleasant odor of wild radish or cabbage
[Fenaroli’s Handbook of Flavor Ingredients. Volume 2. Edited, translated, and revised by T.E. Furia and N. Bellanca. 2nd ed. Cleveland: The Chemical Rubber Co., 1975., p. 406]
pH: not available
Physical form: Colorless Liquid
Refractive Index: 1.4355 @ 20° C/D; MAX ABSORPTION (ALCOHOL): 215 NM (LOG E= 3.0)
[Weast, R.C. (ed.). Handbook of Chemistry and Physics. 60th ed. Boca Raton, Florida: CRC Press Inc., 1979, p. C-505]
Solubility in Water: Max. 2% (w/v) @ 25° C
Specific Heat: @ 20° C Cp (Cal/gm/°C)=0.45
Sulfur Content: 51.5%
Surface Tension: 26.5 dynes/cm= 0.0265 N/m @ 11 deg C 23.33 dynes/cm @ 32. 9° C 19. 87 dynes/cm@ 57. 7° C
[U.S. Coast Guard, Department of Transportation. CHRIS – Hazardous Chemical Data. Manual Two. Washington, DC: U.S. Government Printing Office, Oct., 1978.]
Upper Explosive Limit: 19.7%
Vapor Density: 2.14 (Air= 1)
[Sax, N.I. Dangerous Properties of Industrial Materials. 5th ed. New York: Van Nostrand Rheinhold, 1979., p. 832]
Vapor Pressure @ 20°C (68°F): 530 mbar (398 mm Hg)
Vapor Pressure: 502 mm Hg @ 25° C , from experimentally-derived coefficients.
[Daubert, T.E., R.P. Danner. Physical and Thermodynamic Properties of Pure Chemicals Data Compilation. Washington, D.C.: Taylor and Francis, 1989.]
Viscosity at 25°C (68°F): 0.29 cps
Viscosity: Less than 32.6 Saybolt unit sec
[Clayton, G. D. and F. E. Clayton (eds.). Patty’s Industrial Hygiene and Toxicology: Volume 2A, 2B, 2C: Toxicology. 3rd ed. New York: John Wiley Sons, 1981-1982., p. 2065]
Water Soluble in DMS: Max. 0.4% (w/v) @ 25° C
Wt/vol conversion: 2.54 mg/cu m = 1 ppm
[Clayton, G. D. and F. E. Clayton (eds.). Patty’s Industrial Hygiene and Toxicology: Volume 2A, 2B, 2C: Toxicology. 3rd ed. New York: John Wiley Sons, 1981-1982., p. 2065]
Table 1
Azeotropes Formed with DMS
| Chemical | Percent DMS |
| Methanol | 85 |
| Methyl propyl ether | 65 |
| Isoprene | 35 |
| 2-methyl 2-butene | 52 |
| 2-methyl butane | 15 |
| Pentane | 45 |
| Isopentane | 15 |
| Trimethylethylene | 45 |
Table 2
Solvency Characteristics of DMS
DMS is a nonpolar, stable, water-white organic solvent which is capable of dissolving a wide range of both organic and inorganic materials. In general, DMS will dissolve the following organic materials:
| Material | |
| Alcohols | Ketones |
| Aliphatic hydrocarbons | Monobasic organic acids |
| Amines | Polybutadiene |
| Aromatic hydrocarbons | Polybutylene |
| Ethers | Polyisobutyl methacrylate |
| Esters | Polystyrene |
| Halogenated compounds | Rosins |
The following are not appreciably soluble in DMS:
| Material | |
| Aliphatic dibasic acids | Polyethylene |
| Aromatic dibasic acids | Polyvinyl acetate |
| Dimethyl sulfone | Sulfur |
| Nylon | Urea formaldehyde resins |
Table 3
Solubility of Salts in Dimethyl Sulfide
(Unless otherwise noted, all determinations were made at 25° C)
| Salt | Solubility
grams/100g DMS |
Salt | Solubility
grams/100g DMS |
| Aluminum chloride AlCl3 | > 70g | Chromium fluoride CrF3• 3½H2O | Insoluble |
| Aluminum chloride AlCl3 • 6H2O | Insoluble | Cobalt acetate Co(C2H3O2)2• 4H2O | Insoluble |
| Ammonium bifluoride NH4F•HF | Insoluble | Cobalt chloride CoCl2 | 2.1g at 25°C
26.6g at 50°C |
| Ammonium chloride NH4Cl | Insoluble | Cobalt chloride CoCl2• 6H2O | Insoluble |
| Ammonium chromate (NH4)2CrO4 | Insoluble | Cobaltic oxide Co2O3 | Insoluble |
| Ammonium fluoride NH4F | Insoluble | Columbium pentachloride CbCl5 | very soluble
(evolves heat) |
| Ammonium molybdate (NH4)6Mo7O24• 4H2O | Insoluble | Cupric acetate Cu(C2H3O2)2•H2O | <0.5g |
| Antimony trichloride SbCl3 | ~1200g | Cupric bromide CuBr2 | >10g |
| Arsenic trichloride AsCl3 | Insoluble | Cupric chloride CuCl2• 2H2O | > 20g |
| Arsenic trioxide As2O3 | Insoluble | Cupric sulfate CuSO4• 5H2O | Insoluble |
| Barium chloride BaCl2• 2H2O | Insoluble | Cuprous chloride Cu2Cl2 | 0.31g |
| Bismuth nitrate Bi(N03)3• 5H2O | Insoluble | Cuprous fluoride Cu2F2 | Insoluble |
| Bismuth oxychloride BiOCl | Insoluble | Cuprous iodide Cu2I2 | > 100g |
| Bismuth trichloride BiCl3 | > 125g | Ferric ammonium sulfate NH4Fe(SO4)2• 12H2O | Insoluble |
| Cadmium chloride CdCl2 | 0.24g | Ferric chloride FeCl3 | ~20-30g at 25°C |
| Cadmium iodide CdI2 | 42.3g at 25°C
26.6g at 50°C |
Uranyl acetate UO2(C2H3O2)2• 2H2O | Insoluble |
| Calcium chloride CaCl2 | Insoluble | Vanadium pentoxide V2O5 | Insoluble |
| Chromic oxide Cr203 | Insoluble | Zinc bromide ZnBr2 | 70.0g at 25 °C
203g at 80°C |
| Chromium acetate Cr(C2H3O2)3• 2H2O | Insoluble | Zinc chloride ZnCl2 | ~105 g |
| Chromium Chloride CrCl3• 6H2O | Insoluble | Zinc cyanide Zn(Cn)2 | Insoluble |
Generalizations pertaining to the solubilities of inorganic salts may be misleading and if at all possible, each specific compound should be checked for its solubility.
The halide, cyanide, and acetate salts of the metals occurring in the center of the periodic table tend to be soluble. Alkali and alkaline earth metal salts tend to be insoluble, as do most of the sulfates, carbonates, oxides, and hydrated salts. Some of the metal salts form complexes of varying degree of stability – cuprous cyanide forms a very stable complex, mercuric iodide a very easily decomposed complex.
Table 4
Solubility of Gases in DMS
| Temperature (C°) | Grams of Gas per 100 grams DMS | |||||
| BF3 | BCl3 | HCl | NH3 | H2S | SO2 | |
| 2 | – | – | – | 2.03 | – | – |
| 3 | 114.4 | 39.3 | – | – | – | – |
| 4 | – | – | 27.4 | – | – | – |
| 10 | – | – | – | – | 22.5 | 171.7 |
| 13 | – | – | – | 0.97 | – | – |
| 14 | – | 53.4 | 20.1 | – | – | – |
| 15 | – | – | – | – | 21.8 | 153.0 |
| 16 | 111.5 | – | – | – | – | – |
| 20 | – | – | – | – | 17.1 | 113.0 |
| 25 | – | – | – | 0.45 | 12.5 | 81.2 |
| 26 | 112.0 | 67.5 | 7.6 | – | – | – |
Figure 1
Vapor Pressure-Temperature
Petrochemical
Sulfidation Agent for Olefins Production
DMS is an essential additive in steamcracking operations. When used at trace levels, it serves to control carbon monoxide (CO) generation in the furnace. It also plays a role in managing coke formation rates, which determine furnace run lengths – and ultimately profitable plant operation. DMS is the most widely used sulfidation agent in North America. Gaylord Chemical manufactures a highly pure and consistent DMS product. DMS Exact-S® Grade carefully controls the concentrations of impurities which are of concern to ethylene / propylene manufacturers- notably carbon disulfide.
Ingredient in Gas Odorants
Dimethyl sulfide has a distinctive odor which is detectable in low concentrations. DMS finds use as an odorant in natural gas where it serves as a backup to mercaptan compounds. It is chemically and thermally stable and does not diminish in odor characteristics when exposed to black iron pipe and tanks. The physical properties of DMS make it easy to handle through common odorizing equipment. It boils at 37° C and is volatile enough at the concentrations required that it will not condense in transmission and distribution systems under normal operating temperatures and pressures. Dimethyl sulfide will not fractionate from liquid petroleum gas cylinders nor will it react with compressor oil to form gums, resins, and compounds with low odorizing values.
Fuel Additive
Ethylene oxide containing 1 wt. % dimethyl sulfide may be used as a monopropellant for rocket motors. The use of DMS prevents exhaust nozzle fouling and carbon deposition on the firing chamber surfaces. 5
Extraction Solvent
Dimethyl sulfide, a simple thioether, has a distinct advantage as an extraction solvent in that it does not form dangerous peroxides. In addition, DMS has a low affinity for mineral acids and will not extract these from water solutions. The low water solubility of DMS is another important factor in its use as a replacement for ethers.
Fine Chemical Synthesis
Chemical Reactant
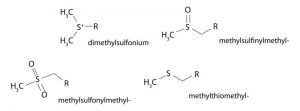
DMS will undergo several types of reactions and may be used as a source for the following reactive groups:
Reaction Medium
Dimethyl sulfide is inert to most reaction components and has a wide range of solubilities for both organic and inorganic materials, including many gases. These characteristics are the basis for its use as a nonaqueous reaction medium. DMS is particularly useful for utilizing soluble gases since lower operating pressures are possible.
Preparation of Carbonyl Cyanide
Carbonyl cyanide can be prepared easily and safely by reacting tetracyanoethylene oxide with DMS. Additional information on this preparation will be found in Bulletin 203B, “Chemical Reactions of Dimethyl Sulfide”.
Catalyst Modifications
The presence of DMS can alter the course of reaction in certain catalytic processes.
A. Selective Hydrogenation
Ethylene which is produced by pyrolysis of natural gas contains varying amounts of acetylene. For most uses of ethylene, the acetylene must be removed. Hydrogenation using a palladium catalyst has been used to accomplish acetylene removal, but this is always accompanied by undesired hydrogenation of ethylene to ethane. When DMS is added to the mixture, it has been found that the hydrogenation of ethylene is suppressed while that of acetylene proceeds normally. The DMS is not decomposed by the treatment and does not have a permanent effect on the catalyst. 3
B. Diolefin Polymerization
Materials consisting of a combination of an organometallic reducing agent and a reducible compound of a heavy metal have been found to serve very well as catalysts for polymerization of diolefins such as butadiene and isoprene. The polymers so produced have a structure predominantly of the cis-1, 4 configuration. While these polymers are very useful for some purposes, for other applications it is desirable to produce polymers having trans-1, 4 configurations. It has been found that such polymers can be produced if the catalyst is modified by the addition of DMS. To insure the 1, 4 configuration, it is also necessary to use iodine either as part of the organometallic reducing agent or as part of the heavy metal reducible compound. 4
Ion Exchange Resins
Strong basic ion exchange resins have been prepared by chloromethylating bead form copolymers of styrene and divinyl benzene followed by reaction with dimethyl sulfide. Treatment with alkali produces a basic resin containing benzyldimethylsulfonium groups.
Modification of Nylon Fibers
A physical and chemical modification of nylon fibers and fabrics may be accomplished by immersion in a solution of DMS and zinc chloride. This treatment will delustre the fibers and make them more receptive to dyeing. It is also claimed that the fiber surfaces will have a higher coefficient of friction, a more rapid moisture pickup, and increased opacity. 1
Fiber Spinning
Polyvinyl chloride polymers can be dry spun or extruded after treatment with DMS and a ketone solvent. Aging of the polymer-solvent mixture produces a viscous solution which can be extruded directly into an atmosphere such that the solvent evaporates readily. Both DMS and the ketone solvent are necessary with their proportions determined by the K value of the polymer. 2
Food and Beverage Component
Dimethyl Sulfide is present above its flavor threshold in most beers. Because of its low flavor threshold, 10 – 150 ppb, it is a primary flavor and aroma compound that makes a significant contribution to beer character, especially in lager beers. Other food applications include non-alcoholic beverages, ice cream, candy, baked goods, gelatins / puddings, and syrups. The use levels are in parts per million.
Dimethyl sulfide is a highly flammable liquid with a flash point of -34°C and an autoignition temperature of 206°C. As a general rule, the same precautions should be employed in the handling of DMS as are used with diethyl ether.
As with any highly flammable material, DMS should be stored in tightly closed containers, in a cool place, and away from strong oxidizing agents or open flames. If operations are to be conducted in closed spaces, explosion-proof electrical switches and motors are suggested, such as will meet specifications of Class 1, Group C, Division 1 of the National Electrical Code. It is also suggested that consideration be given to maintaining an inert atmosphere in the head space when loading or unloading storage tanks or drums.
Storage tanks must be suitable for flammable liquids and may be mild or stainless steel, or aluminum. Nozzles should be flanged and located on top of the tank or vessel whenever possible. Do not expose storage tanks to heat, sparks, or open flames.
Heating coils are not recommended; inadvertent use of heating coils could result in excess vapor generation and tank overpressure. Vents should be adequate to accommodate flow rates during unloading. Vapor controls such as conservation vents, relief valves, floating roofs, scrubbers, carbon absorbers or flares may be required for safety, environmental or odor control purposes. A nitrogen blanket is the preferred atmosphere. Unloading by nitrogen pressure is recommended. Pumping is satisfactory with an inert purge on the transferring vessel and the storage tank. Never use air or oxygen for pressuring.
Piping of mild or stainless steel; flanged construction is recommended for 2” and larger sizes with valves of mild steel, cast iron or stainless steel. Flanges in pipelines carrying DMS should be electri- cally jurnpered so as not to rely on the bolts as conductors. The piping should be designed so that liquid velocities do not exceed 10 feet per second and the entry of DMS into tanks should be at the bottom through a dip tube to avoid static electricity. Do not allow DMS to fall through a vapor space.
Hose of stainless steel wire covering either a Teflon liner or a stainless steel bellows is preferred.
Couplings: Hose fittings should be non-sparking brass or aluminum; an Acme type screw on LPG type fitting is an option.
Gaskets and O-rings should be of Teflon, Gylon. PTFE, or Kalrez. or similar material. Glass-filled TFE and graphite-filled or TFE-filled spiral wound stainless steel gaskets may also be used for piping and equipment flanges.
Pumps may be mild steel or stainless steel with mechanical seals to avoid leaks.
Electrical equipment should be suitable for a Class I hazardous location (Article 500 of the National Electric Code). Specific installations determine whether Division 1 (normally hazardous) or Division 2 (potentially hazardous) location is indicated.
Bibliography
- R. J. Herschler; U. S. Patent 3,023, 074, Feb. 27, 1962.
- F. Siclari and A. Bellano; Italian Patent 508, 513, Jan. 10, 1955.
- L. K. Frevel and L. J. Kressley; U. S. Patent 3,075,024, Jan. 22, 1963.
- R. A. Stewart and E. Lasis; U. S. Patent 3,196,143, July 20, 1965.
- H. B. Welirnan; U. S. Patent 3, 236, 046, Feb. 22, 1966.
The information in this bulletin is based on information available to us and on our observations and experiences. However, no warranty is expressed or implied regarding the accuracy of this data, the results to be obtained from the use thereof, or that any use will not infringe any patent. Each user must establish appropriate procedures for off-loading, handling, and use of the product(s). Since conditions for use are beyond our control, we will make no guarantee of results, and assume no liability for damages incurred by off-loading, handling, or use of the product(s). Nothing herein constitutes permission, or recommendation to practice any invention covered by any patent without license from the owner of the patent.